 |
 |
- Search
| J. People Plants Environ > Volume 26(5); 2023 > Article |
|
ABSTRACT
Background and objective: The aim of this study was to elucidate the effect of varying treatment concentrations of two different plant growth retardants, acting at different inhibition positions during the biosynthetic process, on the growth characteristics of geranium (Pelargonium × hortorum) and then to provide essential data to improve the quality of pot plant production.
Methods: Aqueous solutions of diniconazole (Binnari) and daminozide (B-9) at concentrations of 0 (distilled water), 50, 100, 200, and 400 mg · L−1 were prepared for the geranium ‘Ringo 2000TM Deep Red’. These solutions were applied by foliar spraying twice at 10-day intervals. Growth characteristics such as plant height, leaf size and relative chlorophyll content (SPAD) were then assessed.
Results: When Binnari was applied at concentrations of 0, 50, 100, 200, and 400 mg · L−1, the corresponding plant heights were 29.5, 23.8, 24.1, 23.0, and 18.4 cm, respectively. There was a statistically significant decrease in plant height with increasing concentration. Conversely, for B-9, plant height was significantly reduced to 26.7 cm only at the 400 mg · L−1 concentration, indicating a less pronounced inhibitory effect compared to Binnari. Leaf width showed a significant decreasing trend with increasing Binnari application concentrations and was 4.6, 4.7, 4.1, 4.2, and 3.9 cm respectively. However, there was no significant difference due to the B-9 treatment. Chlorophyll content increased with increasing concentrations of both retardants.
Conclusion: In conclusion, Binnari showed a greater inhibitory effect on geranium growth compared to B-9. The application of Binnari at 400 mg · L−1 resulted in the greatest increase in the ornamental value of the geranium. Furthermore, diniconazole acts during the early stage before the splitting of the GA1 and GA4 pathways, thus inhibiting the biosynthesis of both active gibberellins. In contrast, daminozide inhibits the step just before GA1 biosynthesis. It is concluded that GA4 serves as the dominant active gibberellin in geranium plants.
Methods: Aqueous solutions of diniconazole (Binnari) and daminozide (B-9) at concentrations of 0 (distilled water), 50, 100, 200, and 400 mg · L−1 were prepared for the geranium ‘Ringo 2000TM Deep Red’. These solutions were applied by foliar spraying twice at 10-day intervals. Growth characteristics such as plant height, leaf size and relative chlorophyll content (SPAD) were then assessed.
Results: When Binnari was applied at concentrations of 0, 50, 100, 200, and 400 mg · L−1, the corresponding plant heights were 29.5, 23.8, 24.1, 23.0, and 18.4 cm, respectively. There was a statistically significant decrease in plant height with increasing concentration. Conversely, for B-9, plant height was significantly reduced to 26.7 cm only at the 400 mg · L−1 concentration, indicating a less pronounced inhibitory effect compared to Binnari. Leaf width showed a significant decreasing trend with increasing Binnari application concentrations and was 4.6, 4.7, 4.1, 4.2, and 3.9 cm respectively. However, there was no significant difference due to the B-9 treatment. Chlorophyll content increased with increasing concentrations of both retardants.
Conclusion: In conclusion, Binnari showed a greater inhibitory effect on geranium growth compared to B-9. The application of Binnari at 400 mg · L−1 resulted in the greatest increase in the ornamental value of the geranium. Furthermore, diniconazole acts during the early stage before the splitting of the GA1 and GA4 pathways, thus inhibiting the biosynthesis of both active gibberellins. In contrast, daminozide inhibits the step just before GA1 biosynthesis. It is concluded that GA4 serves as the dominant active gibberellin in geranium plants.
Flowering pot plants are ornamental plants planted in pots to enjoy their flowers, leaves and fruits (KSFS, 2021). Many floriculture plants used for cut flowers tend to have long stems. Even in the case of bedding or garden plants, there are instances where stem elongation can occur, leading to a reduction in quality depending on environmental conditions. In order to grow ornamental plants for cut flower or garden use, it is necessary to inhibit stem elongation, which can be achieved by passive and active methods. Passive methods include techniques such as day-night temperature difference (DIF) and growth inhibitors, while active methods include crossing with dwarf varieties, mutation breeding and genetic transformation (Larson, 1985). Plant growth retardants (PGRs), a type of plant growth regulators, are classified as a category of pesticides along with fungicides, insecticides and herbicides in Korea.
PGRs primarily exert a physiological effect on plants by inhibiting cell division in the apical meristem, thereby reducing stem growth by suppressing active gibberellin (GA) biosynthesis (Bañón et al., 2005). These PGRs have been used extensively in floriculture because of their ability to increase resistance to various environmental stresses such as low temperature, drought, pollution and disease in ornamental plants (Bañón et al., 2005; Bañón et al., 2023). They also contribute to shorter internode lengths, resulting in reduced stem elongation and leaf expansion. They have also been used in cut flower production to maintain quality and improve adaptability to indoor environments (Dennis et al., 1965; Izumi et al., 1984; Kim et al., 2010; Ninnemann et al., 1964).
Commonly used PGRs in agriculture include ancymidol (A-Rest), chlormequat chloride (CCC), chlorphonium chloride (Phosphon), daminozide (B-9), paclobutrazol and prohexidione-calcium (Pro-Ca) (Hashemabadi et al., 2012; Hwang et al., 2009; Kim et al., 2010; Na et al., 2013). In addition, some substances registered as fungicides that also inhibit stem elongation are used as PGRs in crops such as cabbage and turf. Examples include uniconazole, diniconazole, hexaconazole and trinexapac-ethyl (Hashemabadi et al., 2012; Hwang et al., 2009; Kim et al., 2010; Na et al., 2012). Uniconazole, paclobutrazol, cycocel and B-9 have been used in flowering plants. However, the use of B-9 is restricted due to its carcinogenic nature. Substances in the triazole group, such as uniconazole and paclobutrazol, have a wide range of uses in plants (Albrecht and Tayama, 1992; Barret, 1982; Jeong and Kim, 2001; Park et al., 2001). However, the effectiveness of PGRs can vary depending on factors such as type and concentration of treatment, plant species, timing of treatment and method of application (Armitage et al., 1979; Albrecht and Tayama, 1992).
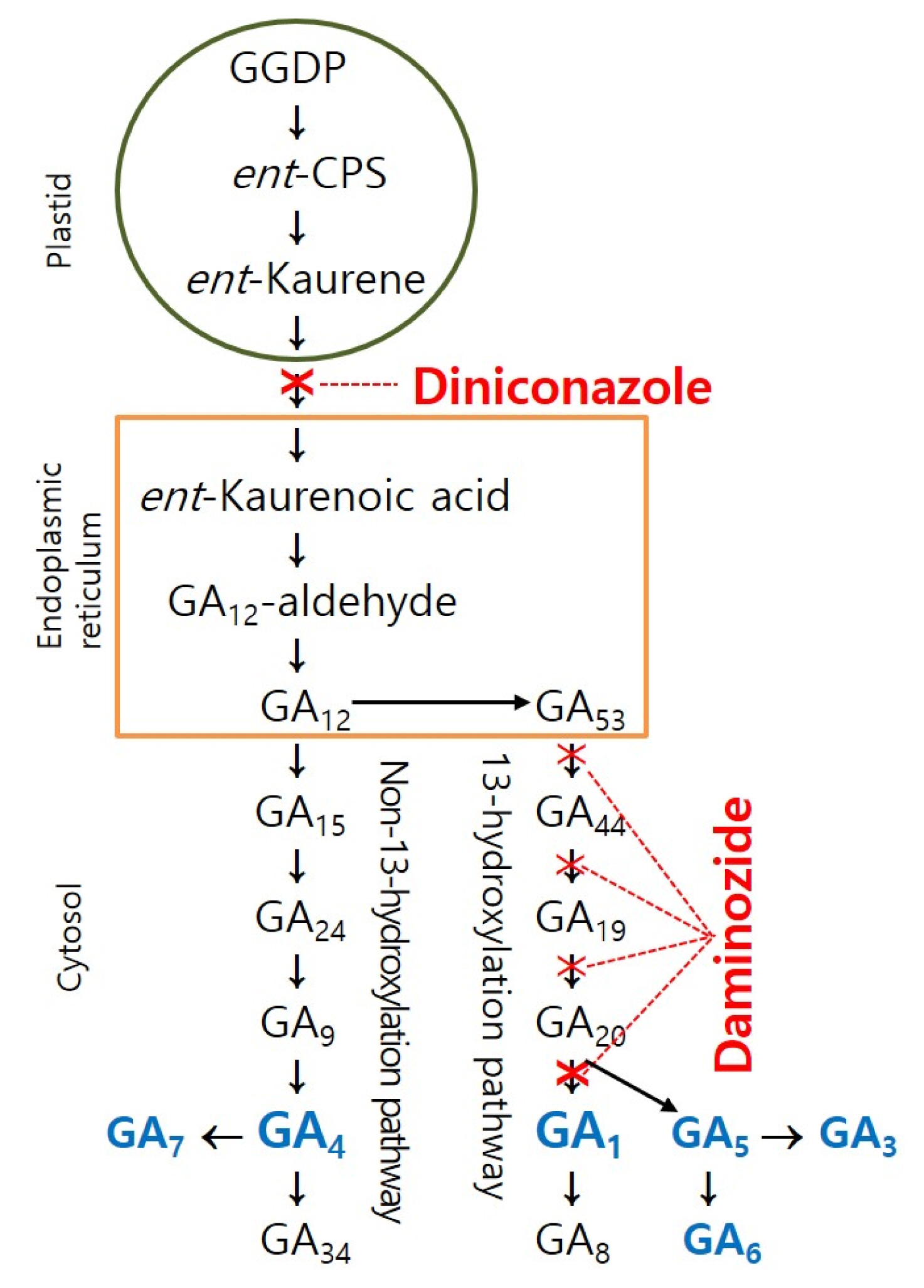
These PGRs interfere with GA biosynthesis processes. CCC primarily inhibits the conversion of geranylgeranyl pyrophosphate to ent-kaurene, while A-Rest, paclobutrazol, uniconazole, diniconazole, and others inhibit the conversion of ent-kaurene to ent-kaurenoic acid (Radermacher, 2016; KSFS, 2021). Subsequently, B-9, Pro-Ca and TE inhibit the conversion of GA53 to the active form GA1.
Geranium (Pelargonium × hortorum) is a tender annual and used as a bedding plant and flowering potted plant (Armitage et al., 1979). Geraniums are often grown at high planting densities to increase productivity, which can lead to excessive stem growth and leaf chlorosis (Albrecht, 1992; Park and Kim, 2001). Therefore, this study investigated the effects of two commercially available PGRs in Korea, diniconazole (Binnari) and daminozide (B-9), on geranium growth. This study aimed to provide basic data to improve the quality of cut flower production by understanding their effects on stem elongation.
The experimental material selected for this study was the geranium (Pelargonium × hortorum) ‘Ringo 2000TM Deep Red’, which was obtained as plug seedlings from ACC Korea-America Seed & Seedling Co., Ltd. (Chungju, Korea). The seedlings were transplanted into 10 cm plastic pots on 19 October and then into 15 cm pots on 30 October. After root establishment, foliar application of PGRs was made on 11 November. The experiment was conducted in a glass greenhouse at Jeju National University (Jeju, Korea). The average greenhouse temperatures during the experiment were 17.3°C in October, 15.7°C in November and 9°C in December. To maintain moisture, overhead irrigation was applied every two days.
Diniconazole is commonly used as a growth retardant in plants to inhibit stem elongation and has been used in crops such as cabbage and turf. Daminozide is widely used to control stem elongation in ornamentals by growers, particularly chrysanthemums, with the advantage of having fewer side effects at higher doses (Davis et al., 1988). Thus, Binnari as a fungicide (a.i. 5% diniconazole, Dongbang Agro, Buyeo, Korea) and B-9 as a growth retardant (a.i. 85% daminozide, Yuwon EcoScience, Iksan, Korea) were selected to investigate the effects of the PGRs on geranium in this study. The recommended appropriate concentration for both Binnari and B-9 in ornamentals is 200 mg · L−1.
To determine concentration effects, solutions of 0, 50, 100, 200, and 400 mg · L−1 were prepared for both Binnari and B-9 using distilled water. The solutions were treated with foliar spray on the plants at 14 mL per plant. The treatments were carried out twice on 11 and 21 November. The control group (0 mg · L−1) was treated with the same volume of distilled water at the same time.
To compare the effects of the PGR treatments, growth characteristics including plant height, leaf length, leaf width, and relative chlorophyll content were measured on 21 November (10 days after the first treatment), 16 December (25 days after the second treatment) and 28 February (after flowering) for six individual plants per treatment. Plant height was measured from the top of the potting medium to the top of the highest stem and leaf width was measured at the widest point of the uppermost mature leaf of each plant. Chlorophyll content was measured using a chlorophyll meter (SPAD-502Plus, Konica Minolta, Japan) targeting an area of approximately 2 mm × 3 mm on the uppermost mature leaf of each plant. Measurements were taken at the same location on each leaf to minimize errors.
In this study, there were three replications per treatment, 10 plants within a replication, and the plants were completely randomized in the benches. Data were analyzed using an ANOVA in SAS software (Window version 9.3, SAS Institute Inc., Cary, NC, USA) to determine significant differences between treatment means using Duncan’s multiple range test (p ≤ 0.05).
In the case of geraniums treated with diniconazole (Binnari) and daminozide (B-9) at concentrations of 50, 100, 200, and 400 mg · L−1, an increasing trend of reduced stem elongation was observed with increasing concentration (Figs. 1A, 1B, 1C). Growth was monitored for 10 days between the first and second measurement periods in the PGR-treated groups. For diniconazole treatment, the newly grown stem lengths, from the 1st measurement to 2nd one, were 2.3, 1.6, 1.6, 1.9, and 1.3 cm in the control group and at concentrations of 50, 100, 200, and 400 mg · L−1, respectively (Figs. 1A, 1B). Similarly, daminozide showed newly grown stem lengths, from the 1st measurement to 2nd one, of 2.0, 1.9, 2.1, and 1.9 cm (Figs. 1A, 1B), indicating a slightly slower growth rate compared to the control group (2.3 cm). After flowering, diniconazole treated plants showed reduced heights of 23.7, 24.1, 23.0, and 18.2 cm at concentrations of 50, 100, 200, and 400 mg · L−1 respectively compared to the control height of 29.5 cm (Fig. 1C). Daminozide treated plants showed reduced heights of 30.8, 27.9, 29.4, and 26.7 cm, with some decreases, but no clear decreasing trend with increasing concentration was observed (Fig. 1C).
Leaf width was smaller in the PGR-treated groups compared to the control, but the effect was shown at lower concentrations in the Binnari (diniconazole) treatment than in B-9 (daminozide). (Figs. 1D, 1E, 1F). In diniconazole-treated plants, the control had a width of 8.5 cm, while concentrations of 50, 100, 200, and 400 mg · L−1 resulted in widths of 7.7, 6.7, 6.3, and 5.6 cm, respectively. However, with daminozide, only the 400 ppm treatment (5.0 cm) showed a significant reduction in leaf width compared to the control (8.5 cm). As a result, the diniconazole treatment was more effective than daminozide in reducing leaf width until the second measurement, before flowering (Figs. 1D, 1E). However, only 400 mg · L−1 diniconazole reduced leaf width after flowering (Fig. 1F).
Regarding the relative chlorophyll content (SPAD) after flowering at different diniconazole concentrations, the control had a SPAD value of 36.2, while concentrations of 50, 100, 200, and 400 mg · L−1 showed values of 41.9, 37.8, 40.0, and 42.2, respectively, with no significant difference at 100 mg · L−1. In contrast, daminozide showed a significant decrease in the SPAD values only at the concentrations of 200 and 400 mg · L−1. Diniconazole treatment showed a proportional increase in relative chlorophyll content with increasing concentration at all three stages (Figs. 1G, 1H, 1I). Daminozide, on the other hand, showed a weaker trend of proportional increase with concentration only in the third measurement (Fig. 1I).
Analysis of the correlation between treatment concentration and stem elongation showed a strong correlation with diniconazole (R2 = 0.81), while a weaker correlation was observed with daminozide (R2 = 0.56) (Figs. 2A, 2B). In addition, both stem elongation and chlorophyll content were significantly affected by the type of PGR (p < .001) and the concentration of PGR (p < .05), but leaf width was not significantly affected (Table 1).
The aim of this study was to investigate the effects of the types and concentrations of PGRs on the growth of geranium in order to improve the quality of geranium production during flowering. Diniconazole (Binnari) and daminozide (B-9), which inhibit different stages of the gibberellin (GA) biosynthesis process (Fig. 3), were used to prepare aqueous solutions at concentrations of 50, 100, 200, and 400 mg · L−1 for foliar application. The results showed that both daminozide and diniconazole at high concentrations reduced stem elongation and increased chlorophyll content in geranium. At high concentrations (400 mg · L−1), diniconazole had a greater inhibitory effect on both stem elongation and leaf width at early growth stages compared to daminozide, and the dominant inhibitory effect of diniconazole was also seen at lower concentrations (Figs. 1 and 2). These findings suggest that diniconazole is more effective than daminozide on ‘Ringo 2000TM Deep Red’ geranium.
GAs are present in almost all higher plants (Lee et al., 2001). Among more than 130 GAs, only a few, including GA1, GA3, GA4, GA5, GA6, and GA7, show biological activity (King et al., 2001, 2003; Yamaguchi, 2008; Hedden and Sponsel, 2015). GA metabolism in higher plants is thought to occur via two major pathways: the early C-13 hydroxylation pathway (GA53 → GA44 → GA19 → GA20 → GA1) and the non-C-13 hydroxylation pathway (GA12 → GA15 → GA24 → GA9 → GA4) (Sponsel, 1995). Different plant species have different levels and relative amounts of active GAs, with GA1 and GA4 identified as regulators of stem elongation (Oh et al., 2015). Diniconazole, an active component of diniconazole, acts as a triazole-based GA biosynthesis inhibitor, targeting an intermediate step in the pathway and inhibiting both with GA1 and GA4 biosynthesis prior to their divergence. In contrast, daminozide, the active component of daminozide, inhibits the pathway just before the formation of active GA1 (Fig. 3). This suggests that GA4 or GA7 is the dominant active GA in geranium.
Another experiment comparing the effects of diniconazole at concentrations of 100, 200 and 400 mg · L−1, commonly used as a fungicide, on daylily and sage plants showed that stem growth was suppressed with increasing concentration (Choi et al., 2011). In our study, both diniconazole and daminozide at a high concentration of 400 mg · L−1 resulted in shorter stem elongation, reduced leaf width and increased chlorophyll content. This suggests that both diniconazole and daminozide at 400 mg · L−1 are effective in reducing stem elongation to improve the quality of flowering plants.
While PGRs can increase chlorophyll content in ornamental plants (Wang and Gregg, 1990), the results of the diniconazole treated groups did not show significant differences in chlorophyll content. However, in the daminozide treated groups, higher concentrations resulted in higher chlorophyll content.
In conclusion, Binnari had a more pronounced inhibitory effect on geranium growth than B-9. Application of Binnari at 400 mg · L−1 resulted in the greatest increase in the ornamental value of the geranium. Furthermore, diniconazole acts during the early stage before the cleavage of the GA1 and GA4 pathways, thus inhibiting the biosynthesis of both active gibberellins. In contrast, daminozide inhibits the step just before GA1 biosynthesis. This suggests that GA4 is the dominant active gibberellin in geranium plants.
Fig. 1
Plant height (A–C), leaf width (D–F), and chlorophyll content (G–I) of Pelargonium × hortorum ‘Ringo 2000™ Deep Red’ as influenced by foliar application of 0, 50, 100, 200, 400 mg · L−1 Binnari (diniconazole) and B-9 (daminozide). The 1st measurement (A, D, G), the 2nd measurement (B, E, H), and the 3rd measurement (C, F, I). Within-graph means followed by the same letter are not significantly different by Duncan’s multiple range test at p ≤ 0.05.

Fig. 2
Relationship between concentration of plant growth retardants and plant height in Pelargonium × hortorum ‘Ringo 2000™ Deep Red’ as influenced by foliar application of 0, 50, 100, 200, and 400 mg · L−1 Binnari (diniconazole) and B-9 (daminozide). Error bars indicate SE (n = 3).

Fig. 3
Schematic diagram of the biosynthesis of gibberellins and the steps blocked by diniconazole and daminozide. GA1 and GA4 are known to be biologically active including GA3, GA5, GA6, and GA7. This diagram is adapted from Katyayini et al. (2020) and Radermacher (2016).
References
Albrecht, M.L., H.K. Tayama. 1992. Tips on the use of chemical growth regulators on floriculture crops Ohio Florists’ Association. Columbus, OH, USA:
Armitage, A.M., M.J. Jusjita, P.J. Harney. 1979. Effect of Cycocel and high intensity lighting on flowering of seed propagated geranium. Journal of Horticultural Science. 53:147-149.
https://doi.org/10.1080/00221589.1978.11514810

Bañón, S., J.A. Fernández, J. Ochoa, M.J. Sánchez-Blanco. 2005. Paclobutrazol as an aid to reduce some effects of salt stress in oleander seedlings. European Journal of Horticultural Science. 70:43-49.
Bañón, D., M.F. Ortuño, M.J. Sánchez-Blanco, B.L. Pagán, S. Bañón. 2023. Effects of paclobutrazol and mepiquat chloride on the physiological, nutritional, and morphological behavior of potted Icterina sage under greenhouse conditions. Agronomy. 13(8):2161.
https://doi.org/10.3390/agronomy13082161

Barret, J.E. 1982. Chrysanthemum height control by ancymidol, PP333 and EL-500 dependent on medium composition. HortScience. 17:896-897.
https://doi.org/10.21273/HORTSCI.17.6.896

Choi, S.H., J.-S. Kang, Y.-W. Choi, Y.-J. Lee, Y.-H. Park, M.-R. Kim, B.-G. Son, H.-K. Kim, H.-Y. Kim, W. Oh, H.-B. Sim, K.-B. Lim, J.-K. Kim. 2011. Effect of diniconazole on growth and flowering of Vinca rocea and Salvia splendis
. Journal of Life Science. 21:1004-1008.

Davis, T.D., G.L. Stefferns, N. Sankhla. 1988. Triazole plant growth regulators. Horticultural Review. 10:63-105.
https://doi.org/10.1002/9781118060834

Dennis, D.T., C.D. Upper, C.A. West. 1965. An enzymic site of inhibition of gibberellin biosynthesis by Amo 1618 and other plant growth retardants. Plant Physiology. 40:948.
https://doi.org/10.1104/pp.40.5.948



Eum, S.J., K.I. Park, I.J. Lee, Y.J. Choi, W. Oh, K.W. Kim. 2011. Effects of foliar-sprayed diniconazole on contents of endogenous gibberellic acids and abscisic acid in Lilium dauricum
. Korean Journal of Horticultural Science and Technology. 29(3):165-171.
Hashemabadi, D., S.R. Lipaei, V. Shadparvar, M. Zarchini, B. Kaviani. 2012. The effect of cycocel and daminozide on some growth and flowering characteristics of Calendula officinalis L., an ornamental and medicinal plant. Journal of Medicinal Plants Research. 6:1752-1757.
https://doi.org/10.5897/JMPR011.1561

Hwang, I.C., I.J. Lee, T.K. Cho, J.T. Kim, C.S. Yoon. 2009. Development of laborsaving plant growth regulator, prohexadione-calcium, for growth inhibition. Korean Journal of Weed Science. 29:1-8.
Izumi, K., I. Yamaguchi, A. Wada, H. Oshio, N. Takahashi. 1984. Effects of a new plant growth retardant (E)-1-(4-chlorophenyl)-4, 4-dimethyl-2-(1, 2, 4-triazol-1-yl)-1-penten3-ol (S-3307) on the growth and gibberellin content of rice plants. Plant and Cell Physiology. 25:611-617.
https://doi.org/10.1093/oxfordjournals.pcp.a076751

Jeong, J.H., G.H. Kim. 2001. Effects of cutting time, pinching and growth regulators on growth and flowering of Sedum spectabile and Sedum atzoon
. Journal of Korea Flower Research Society. 9:59-66.
Katyayini, N.U., P.L. Rinne, D. Tarkowská, M. Strnad, C. van der Schoot. 2020. Dual role of gibberellin in perennial shoot branching: Inhibition and activation. Frontiers in Plant Science. 11:736.
https://doi.org/10.3389/fpls.2020.00736



Kim, Y.H., A.L. Khan, M. Hamayun, J.T. Kim, J.H. Lee, I.C. Hwang, C.S. Yoon, I.J. Lee. 2010. Effects of prohexadione calcium on growth and gibberellins contents of Chrysanthemum morifolium R. cv. Monalisa White. Scientia Horticulturae. 123:423-427.
https://doi.org/10.1016/j.scienta.2009.09.022

Korean Society for Floricultural Science (KSFS). 2021. Floriculture World Science. Seoul, Korea: (pp. 220 p.
Na, C.I., M. Hamayun, A.L. Khan, Y.H. Kim, K.I. Choi, S.M. Kang, S.I. Kim, J.T. Kim, J.G. Won, I.J. Lee. 2013. Influence of prohexadione-calcium, trinexapac-ethyl and hexaconazole on lodging characteristic and gibberellin biosynthesis of rice (Oryza sativa L.). African Journal of Biotechnology. 10:13097-13106.
Ninnemann, H., J.A. Zeevaart, H. Kende, A. Lang. 1964. The plant growth retardant CCC as inhibitor of gibberellin biosynthesis in Fusarium moniliforme
. Planta. 61:229-235.


OH, W., J.S. Kim, Y.H. Kim, I.J. Lee, K.S. Kim. 2015. Shoot elongation and gibberellin contents in Cyclamen persicum are influenced by temperature and light intensity. Horticulture, Environment, and Biotechnology. 56(6):762-768.
https://doi.org/10.1007/s13580-015-1124-8

Park, B.M., S.K. Kim. 2001. Effect of folia application growth retardants on the growth and flowering of geranium ‘Pinto Scarlet’. Journal of Korean Flower Research Society. 9:9-12.
Radermacher, W. 2016. Chemical regulators of gibberellin status and their application in plant production. In: Hedden P., Thomas S.G., (Eds), The gibberellins. Annual Plant Review 49:(pp. 359-403).
https://doi.org/10.1002/9781119210436.ch12

Wang, Y.T., L.L. Gregg. 1990. Uniconazole affects vegetative growth flowering, and stem anatomy of Hibicus
. Journal of American Society for Horticultural Science. 114:927-932.
https://doi.org/10.21273/JASHS.114.6.927








